Health Assessment & Technology Evaluation Market to Reach $3.3B by 2033 at 15.8% CAGR Driven by Gene Therapy Innovation
Gene therapy breakthroughs and precision medicine are accelerating the Health Assessment & Technology Evaluation Market toward $3.3B by 2033 at 15.8% CAGR.
Precision medicine, AI diagnostics, and gene therapy advances are driving the Health Assessment & Technology Evaluation Market to $3.3Billion by 2033.”
AUSTIN, TX, UNITED STATES, February 27, 2026 /EINPresswire.com/ -- The Health Assessment and Technology Evaluation Market is emerging as a cornerstone of modern healthcare systems, enabling governments, payers, and healthcare providers to make informed decisions regarding the adoption, reimbursement, and utilization of medical technologies. Health Technology Assessment (HTA) integrates clinical effectiveness, cost-effectiveness, safety, ethical considerations, and social impact to evaluate pharmaceuticals, medical devices, diagnostics, and digital health innovations. As healthcare costs rise globally and new high-value therapies enter the market, structured health assessment frameworks are becoming essential for ensuring sustainability and equitable access to care.— DataM Intelligence
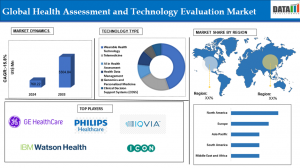
According to DataM Intelligence, the Global Health Assessment and Technology Evaluation Market reached US$ 760.22 million in 2024 and is expected to reach US$ 3,304.84 million by 2033, growing at a CAGR of 15.8% during the forecast period 2025–2033. The strong double-digit growth reflects increasing demand for value-based healthcare models, rising healthcare expenditures, and regulatory mandates for economic evaluation before reimbursement approvals. The pharmaceutical and biotechnology segment leads the market due to the expanding pipeline of high-cost biologics, specialty drugs, and gene therapies that require detailed cost-utility and budget impact analyses. Geographically, North America dominates the market, supported by advanced regulatory infrastructure, established reimbursement systems, and strong adoption of health economics and outcomes research (HEOR) practices.
To Download Sample Report Here: https://www.datamintelligence.com/download-sample/health-assessment-and-technology-evaluation-market
Key Highlights from the Report:
➤ The market is projected to grow at a CAGR of 15.8% between 2025 and 2033.
➤ Pharmaceuticals and biotechnology represent the leading technology segment due to increasing reimbursement scrutiny.
➤ Economic evaluation services hold the largest service share within the market.
➤ North America accounts for the highest revenue contribution globally.
➤ Growing adoption of real-world evidence (RWE) is enhancing technology evaluation frameworks.
➤ Emerging economies are increasingly integrating structured health technology assessment processes into healthcare policy decisions.
Recent Developments :
October 2025: National Institute for Health and Care Excellence launched an accelerated digital health assessment framework to fast-track the evaluation of AI-driven diagnostic platforms and remote patient monitoring tools. The initiative aims to shorten appraisal timelines while maintaining rigorous clinical and economic evaluation standards, reflecting the growing integration of digital therapeutics and software-as-a-medical-device (SaMD) solutions.
September 2025: Institute for Clinical and Economic Review introduced an updated value assessment framework incorporating real-world evidence (RWE) and health equity metrics into cost-effectiveness analyses. The revision strengthens payer decision-making processes by aligning reimbursement models with long-term population health outcomes.
August 2025: Canadian Agency for Drugs and Technologies in Health expanded its health technology assessment (HTA) scope to include advanced cell and gene therapies. The agency implemented adaptive reimbursement models and lifecycle-based evaluations to address high upfront treatment costs and long-term clinical uncertainties.
June 2025: World Health Organization released updated global guidance on institutionalizing HTA in low- and middle-income countries. The framework supports evidence-based priority setting, universal health coverage (UHC) goals, and sustainable healthcare financing strategies.
Mergers & Acquisitions:
November 2025: A leading global health economics consultancy acquired a real-world data analytics firm to strengthen capabilities in outcomes research, pharmacoeconomics, and payer evidence generation for innovative biologics and specialty drugs.
September 2025: A multinational healthcare advisory company completed the acquisition of a digital evidence modeling startup specializing in AI-powered cost-effectiveness simulation platforms, enhancing technology evaluation efficiency and predictive analytics capabilities.
July 2025: A prominent clinical research organization expanded its market access division by acquiring a boutique HTA advisory firm, enabling integrated services spanning clinical trials, value dossier development, and reimbursement strategy.
May 2025: A global life sciences data company acquired a health outcomes research and policy consulting group to enhance its value-based healthcare portfolio, supporting payers and providers in technology adoption and reimbursement decision-making.
Buy Now & Unlock 360° Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=health-assessment-and-technology-evaluation-market
Company Insights
Key players operating in the Health Assessment and Technology Evaluation Market include:
Philips Healthcare
GE Healthcare
IQVIA
IBM Watson Health
ICON Plc
Omron Healthcare
Orcha Health
Emerging Market Players:
AliveCor
Tempus Labs
Health Catalyst
Orpyx Medical Technologies
Freenome
Biofourmis
Virta Health
Market Segmentation Analysis:
The Health Assessment and Technology Evaluation Market is segmented by service type, technology type, end-user, and application.
By service type, the market includes economic evaluation, clinical effectiveness assessment, safety and risk-benefit analysis, budget impact analysis, systematic reviews, and real-world evidence studies. Among these, economic evaluation services dominate due to their critical role in determining incremental cost-effectiveness ratios (ICER), quality-adjusted life years (QALYs), and cost-benefit thresholds that influence reimbursement decisions.
Based on technology type, the market encompasses pharmaceuticals, medical devices, in-vitro diagnostics, digital health technologies, and surgical interventions. The pharmaceutical segment leads the market, driven by increasing regulatory requirements for value demonstration prior to market access. With rising innovation in oncology, immunotherapy, and rare disease treatments, companies are increasingly investing in health technology assessment services to support pricing negotiations and market access strategies.
By end-user, the market serves government agencies, healthcare providers, insurance companies, pharmaceutical and biotechnology companies, and academic research organizations. Government agencies represent a significant portion of the market, as national health authorities rely heavily on HTA frameworks for formulary listing and policy-making decisions. Pharmaceutical and biotechnology firms are also key contributors, leveraging HEOR studies to enhance product commercialization and global market entry strategies.
In terms of application, the market includes reimbursement decision-making, policy development, clinical guideline formulation, healthcare investment prioritization, and pricing strategy optimization. Reimbursement support remains the leading application, reflecting the growing importance of cost containment and value-based pricing models in healthcare systems worldwide.
Regional Insights:
North America holds the dominant position in the Health Assessment and Technology Evaluation Market due to strong institutional frameworks, advanced healthcare infrastructure, and high healthcare spending. The United States and Canada have mature systems for evaluating cost-effectiveness and clinical value, supporting widespread adoption of HTA services. The shift toward outcome-based reimbursement models and accountable care organizations further fuels market growth in the region.
Europe follows closely, driven by established national HTA agencies and harmonized regulatory approaches across member states. Countries such as Germany, the United Kingdom, and France integrate health technology assessment into reimbursement and pricing negotiations. Europe’s focus on universal healthcare access and cost control strengthens demand for comprehensive technology evaluation services.
The Asia-Pacific region is witnessing rapid growth, supported by healthcare system modernization, expanding insurance coverage, and increasing awareness of economic evaluation methods. Countries such as China, Japan, South Korea, and India are progressively adopting structured HTA mechanisms to guide policy and reimbursement decisions. Growing investments in healthcare infrastructure and pharmaceutical R&D further enhance regional prospects.
Latin America and the Middle East & Africa are gradually adopting HTA frameworks, though at varying levels of maturity. Healthcare reforms, increasing public health expenditure, and international collaborations are expected to create growth opportunities in these emerging markets.
Market Dynamics:
Market Drivers
A primary driver of the Health Assessment and Technology Evaluation Market is the rising global healthcare expenditure coupled with cost containment pressures. As innovative but expensive treatments enter the market, payers require evidence of clinical and economic value before approving reimbursement. Health technology assessment provides a structured approach to evaluate long-term outcomes, comparative effectiveness, and budget impact.
The increasing burden of chronic diseases such as cardiovascular disorders, diabetes, cancer, and neurological conditions also drives demand for systematic evaluation of treatment options. Policymakers seek cost-effective solutions to manage growing patient populations without compromising quality of care.
Additionally, the growing adoption of value-based healthcare models significantly contributes to market expansion. Payment systems are increasingly linked to patient outcomes rather than service volume, making robust technology evaluation critical for performance measurement and resource allocation.
Market Restraints
Despite strong growth prospects, the market faces certain limitations. One key restraint is the lack of standardized methodologies across countries, which creates inconsistencies in cost-effectiveness thresholds and reimbursement decisions. Multinational companies often encounter regulatory complexity when navigating diverse HTA frameworks.
Another challenge is limited data availability in emerging markets. Reliable real-world evidence and long-term outcome data are essential for comprehensive assessments, but infrastructure gaps and fragmented health information systems can hinder evaluation quality.
The time-intensive nature of technology evaluation processes may also delay product approvals and market access, particularly for small and mid-sized healthcare companies with limited resources.
Market Opportunities
Significant opportunities lie in the integration of advanced analytics, artificial intelligence, and big data into HTA methodologies. The growing use of electronic health records and digital health platforms provides extensive datasets for real-world evidence generation, improving the accuracy of cost-effectiveness analysis.
Emerging economies represent substantial untapped potential as governments introduce formal reimbursement policies and health financing reforms. Consulting firms and specialized HTA service providers can expand operations by supporting these regions in building standardized evaluation frameworks.
Collaborations between academia, regulatory agencies, and industry stakeholders also offer opportunities to enhance transparency, improve methodological consistency, and accelerate technology adoption while maintaining economic sustainability.
Get Customization in the report as per your requirements:https://www.datamintelligence.com/customize/health-assessment-and-technology-evaluation-market
Frequently Asked Questions (FAQs):
◆ How Big is the Health Assessment and Technology Evaluation Market in 2024?
◆ What is the Expected CAGR of the Health Assessment and Technology Evaluation Market during 2025–2033?
◆ Who are the Key Players in the Global Health Technology Assessment Market?
◆ What is the Market Forecast for the Health Assessment and Technology Evaluation Market by 2033?
◆ Which Region is Estimated to Dominate the Health Assessment and Technology Evaluation Industry through the Forecast Period?
Conclusion:
The Health Assessment and Technology Evaluation Market is experiencing rapid growth, driven by escalating healthcare costs, regulatory reforms, and the global shift toward value-based care. With the market projected to grow from US$ 760.22 million in 2024 to US$ 3,304.84 million by 2033 at a CAGR of 15.8%, the sector is poised for substantial expansion. Pharmaceutical innovation, digital health advancements, and increasing demand for cost-effectiveness analysis are reinforcing the importance of structured health technology assessment frameworks. Although challenges such as data limitations and regulatory variability remain, advancements in analytics and global healthcare reforms are expected to create sustained growth opportunities in the coming years.
Related Reports:
E-health Market
MHealth Market
Sai Kiran
DataM Intelligence 4Market Research
+1 877-441-4866
email us here
Visit us on social media:
LinkedIn
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.